Electrocatalytic Materials Research
NLR's materials discovery and design work on electrocatalytic materials focuses on ceramic proton conductive fuel cells and electrolysis materials, including proton exchange membrane electrolysis catalysts.
Solid-Oxide Electrolysis Cell Materials
We develop materials for intermediate temperature proton-conducting solid-oxide electrolysis cell that can electrochemically generate hydrogen at elevated temperatures. The high-throughput screening methods involve combinatorial thin-film deposition and spatially resolved characterization of the resulting materials. Studies also include evaluation of electrochemical performance and grain boundary diffusion.
For more information, see:
Combinatorial Impedance Spectroscopy with Bayesian Analysis for Triple Ionic-Electronic Conducting Perovskites, Journal of Materials Chemistry A (2022)
Rapid Mapping of Electrochemical Processes in Energy-Conversion Devices, Joule (2024)
Triple Ionic–Electronic Conducting Oxides for Next-Generation Electrochemical Devices, Nature Materials (2021).
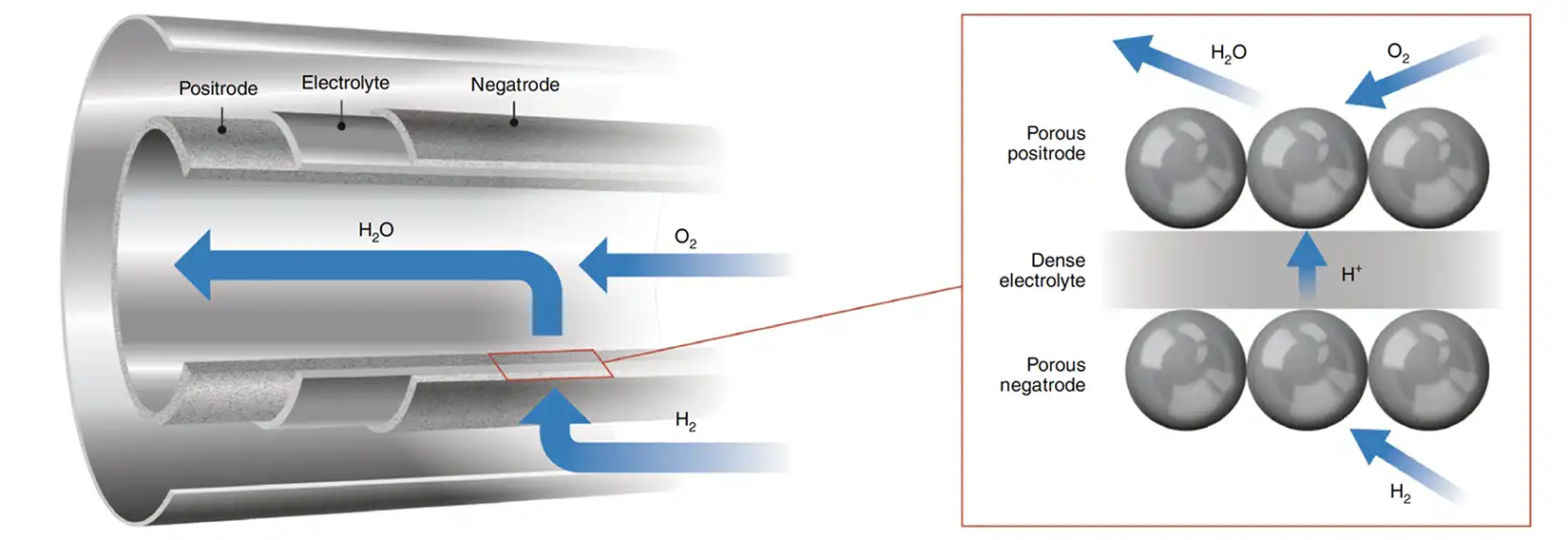
Proton Exchange Membrane Electrolysis Materials
Our electrolysis materials research pursues platinum group metal-free catalysts to reduce the cost of proton exchange membrane fuel cells while retaining high performance. Understanding catalysts with a heterogenous structure is limited by information depth and spatial resolution of common characterization techniques. We use sputtered thin films as a promising model system to study the oxygen evolution reaction active sites, in conjunction with surface analysis and electrocatalytic performance measurements, because of their potentially smooth and homogenous surface.
For more information, see X-Ray Photoelectron Spectroscopy and Rotating Disk Electrode Measurements of Smooth Sputtered Fe-N-C Films, Applied Surface Science (2020).
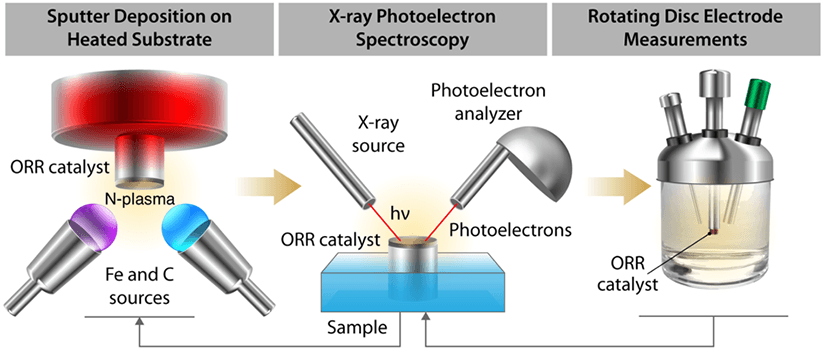
Projects
NLR's work on electrocatalytic materials research for hydrogen production and utilization is funded by the U.S. Department of Energy (DOE) and DOE's Office of Science, Basic Energy Sciences.
HydroGEN is a consortium of six DOE national laboratories that will address advanced water-splitting materials challenges by making unique, world-class national lab capabilities in photoelectrochemical, solar thermochemical, and low- and high-temperature electrolytic water splitting more accessible to academia, industry, and other national labs.
ElectroCat's national laboratory partners advance the tools needed to model, characterize, and optimize platinum group metal-free -catalysts and electrode structures to the point that they are easily applied to a broad range of catalyst systems and set a standard for rapid material analysis.
Research Collaborators
Ryan O’Hayre – Bulk oxides (Colorado School of Mines)
Sossina Haile – Epitaxial oxide materials (Northwestern)
Contacts
Share
Last Updated Dec. 7, 2025
