Scientists Pinpoint Pathways for Carbon Dioxide Uptake of Cyanobacteria
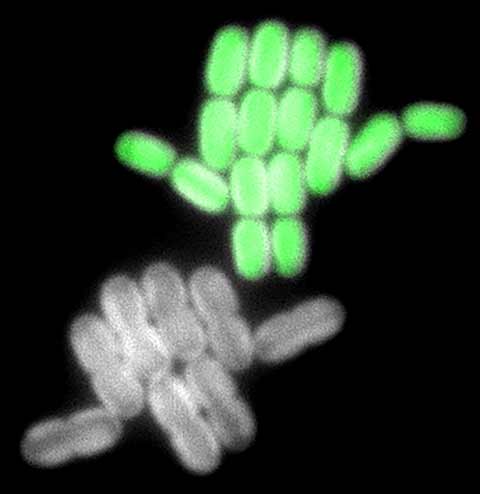
But bioscientists have faced a sizeable research roadblock. They have been unable to accurately measure the growth of individual cyanobacteria cells, which is key to understanding the mechanisms by which they fix CO2 efficiently.
Thanks to research by scientists with the Renewable and Sustainable Energy Institute (RASEI)—a joint institute between the National Renewable Energy Laboratory (NREL) and the University of Colorado Boulder—we can now do just that.
Research published in two separate articles shows how RASEI researchers traced the growth of individual cyanobacteria, providing new insights into the biological pathways that enable efficient photosynthetic growth.
“This is the first time tracking a single [carboxysome] protein complex over multiple generations,” said Jeffrey Cameron, an RASEI researcher and coauthor of the Science Advances article, “Life Cycle of a Cyanobacterial Carboxysome.”
Carboxysomes have been known to be the site of carbon fixation in cyanobacteria. But Cameron and his colleagues wanted to understand the relationship between carboxysome activity and cell growth.
To do this, they used synthetic biology to generate cells with single carboxysomes and filmed them over 60 hours to see how they responded to varying levels of CO2. As the cells grew and expanded, the researchers were able to trace the carboxysomes as they were passed down from one generation to the next.
As a result, they were able to see what conditions and mechanisms enabled highly productive cell growth. Those cells without carboxysomes stopped growing completely, unable to take CO2 from the air. But other cells with carboxysomes showed remarkable growth and reproduction, passing on their CO2-fixing talent to their children.
“We classified 5% of the carboxysomes as ultra-productive,” Cameron said. “If we could get all of them to be like this, we might significantly enhance carbon fixation.”
A Preliminary Problem: Measuring a Single Cell
According to Cameron, however, the ability to follow carboxysomes across multiple generations first required overcoming a practical hurdle: growing cells evenly.
Scientists have traditionally used large cyanobacteria cultures with billions of cells to study light absorption. But Cameron explained that the cells in such experiments shade each other, creating an uneven climate whereby some cells receive more light than others. To understand carbon fixation, they first needed to create an experiment to measure it.
“You are blinded when you try to average an entire population of cyanobacteria, so we wanted to develop a framework for single cell analysis,” he said.
As detailed in the Nature Microbiology article, “Mechanical Regulation of Photosynthesis in Cyanobacteria,” Cameron and his colleagues achieved unprecedented precision by growing cells in a single layer. They could then control the amount of light each cell received and observe how they reacted.
“We turned the light on, and they grew completely identically because we had such control,” Cameron said.
With such nimbleness, the researchers were able to watch the growth of individual cells in a colony, which grew and bathed in the light until they became squished together. Instead of pushing neighbor cells out of the way or bursting, the cyanobacteria simply dissipated the energy, becoming “highly fluorescent,” according to Cameron.
By noting this response, the researchers were able to refine their experiment design to allow each cell to complete its growth cycle.
A Pathway to Efficient Bioenergy?
The findings from these two experiments have the potential to fuel the development of bioenergy technologies.
“It’s been really challenging to produce renewable fuels from cyanobacteria because of limited yield,” Cameron said. “Our single cell experiments can help understand the real limitations and bottlenecks for photosynthetic production of high-value fuels and chemicals.”
This might involve introducing carboxysomes into plants and other single-celled organisms, allowing engineers to dramatically improve the efficiency of carbon fixation. Cameron points to multiple applications for this: more productive biofuel crops like switchgrass or ultra-efficient bioreactions that turn sunlight into valuable chemicals.
What seems clear from Cameron’s research is that the admirable properties of cyanobacteria may hold the keys for making these novel biotechnologies a reality.
These studies were supported, in part, by the National Science Foundation, Oak Ridge Institute for Science and Education, a National Institutes of Health T32 training grant, the U.S. Department of Energy, and a RASEI seed grant.
Learn more about bioenergy research at NREL.
Last Updated Jan. 22, 2026
