Semiconductor Electrolyte Interfaces
NREL examines how interfacial chemistry and microenvironments affect the thermodynamics and kinetics of charge separation and redox properties at (photo)electrocatalyst-electrolyte interfaces of semiconductors.
The goal is to understand how surface modification, phase, defects, surface (re)construction, and co-catalysts in semiconductor-electrolyte systems influence behavior under realistic conditions.
Carbon Dioxide and Nitrogen Reduction Reactions Using 2D Transition Metal Dichalcogenide and Carbide/Nitride Catalysts
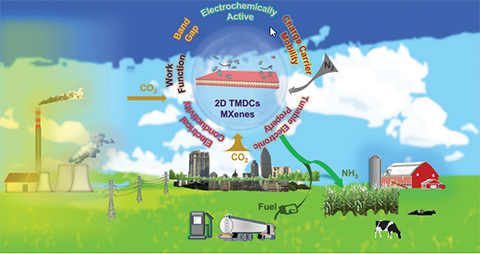
Carbon Dioxide and Nitrogen Reduction Reactions Using 2D TMDC and MXene Catalysts, Energy & Environmental Science (2021)
With an eye towards next-generation technologies with lower energetic costs and greenhouse gas emissions, we addressed (photo)electrocatalytic and photocatalytic trends, and lessons learned to improve 2D transition metal dichalcogenide (TMDC) and transition metal nitrides/carbide (MXene) catalysts for carbon dioxide and nitrogen reduction reactions.
Research Details
- Explored 2D TMDC and MXene materials for carbon dioxide and nitrogen reduction reactions
- Addressed how defects, phases, edge sites, interfaces, doping, and functional groups can be engineered to improve catalytic performance
- Provided our perspective on advancing these two material classes within (photo)electrocatalytic and photocatalytic setups before they could be considered industrially viable catalysts.
Significance and Impact
Converting carbon dioxide into useful C-based products and revolutionizing industrial ammonia generation are two scientific grand challenges with the potential to solve critical global energy and greenhouse gas threats. We explored (photo)electrocatalytic and photocatalytic methods as potential solutions by addressing 2D TMDC and MXene catalysts.
Plasmonic Hot-Hole Transfer in Gold Nanoparticle-Decorated Transition Metal Dichalcogenide Nanosheets
Transient absorbance measurements showed that molybdenum diselenide (MoSe2) and tungsten diselenide (WSe2) decorated with gold nanoparticles show hot-hole transfer from the gold to the transition metal dichalcogenide, producing a charge-separated state.
Research Details
- Gold nanoparticle-decorated mono- to few-layer transition-metal dichalcogenides nanosheets were prepared using liquid-phase exfoliation and direct growth of gold nanoparticles from solution.
- Thin films were deposited by blade coating.
- Photogenerated carrier dynamics in these nanosheets were studied using transient absorbance.
Significance and Impact
Efficient injection of hot holes at the metal-semiconductor interface represents an important advance in harnessing plasmonic dissipation pathways to generate long-lived carriers suited for solar-driven light harvesting and catalysis.
Partner
Scanning Eletrochemical Microscopy Reveals Hydrogen Evolution Reaction and Semiconducting Properties of MXene
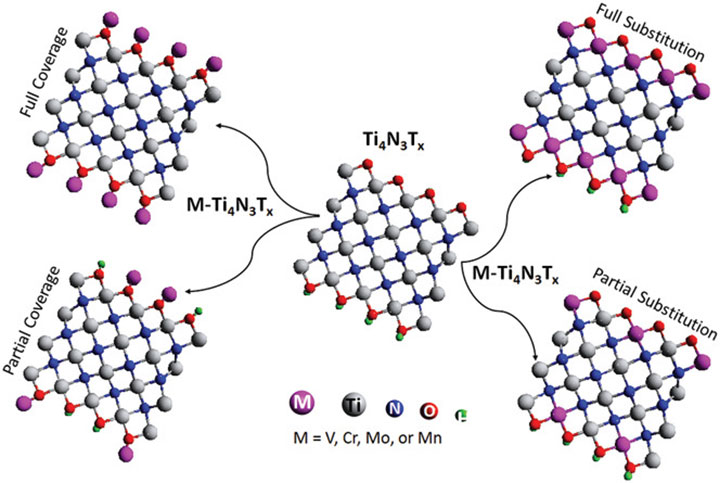
Basal Plane Hydrogen Evolution Activity from Mixed Metal Nitride MXenes Measured by Scanning Electrochemical Microscopy, Advanced Functional Materials (2020)
2D mixed-metal nitride MXenes M-Ti4N3Tx (M = V, Cr, Mo, Mn) were synthesized by a novel solution-based method and characterized via scanning electrochemical microscopy.
Research Details
Hydrogen evolution reaction activity results from the transition metal ions comprising the outer layer of the basal plane in MXenes, which is distinct from other 2D materials such as transition metal dichalcogenides where the catalytically active transition metals are only exposed on the edges.
There is much debate in the community surrounding whether MXenes are intrinsically metals or can exhibit semiconducting properties, and this report conclusively demonstrates the latter.
Significance and Impact
Scanning electrochemical microscopy data showed the first experimental evidence that MXenes exhibit hydrogen evolution reactionactivity from their basal planes, the large area "flat" part of the 2D structure. Additionally, scanning electrochemical microscopy provided evidence for semiconducting behavior, which represents only a handful of reports showing that MXenes can be semiconducting in addition to metallic.
Partner
Contact
Share

